|
For example, a quick glance at Figure AT5.2. That way, we can get a better look at the relationship. These atoms are often called the alkali and alkaline earth elements. For example, potassium has a configuration Ar4s 1. 1 ), the configuration is a closed shell of core electrons, plus s electrons in a new shell. Often it is useful to plot data on a graph. For atoms found in the first two columns of the periodic table (figure 1.6.1 1.6. As a result, fluorine is usually thought of as the most electronegative element. B: 1s 2 2s 2 2p 1 (there are three electrons on the highest occupied energy level n2) In fact, the number of valence electrons goes up by one for each step across a. In addition, the total number of electron shells an atom has determines which period it belongs to. However, on many scales, fluorine would be the most electronegative atom here. How many valence electrons does boron have Recognize that the second principal energy level consists of both the (2s) and the (2p) sublevels, and so the answer is three. An element’s location in the periodic table is largely dependent on its electrons the number of valence shell electrons determines its group, and the type of orbital in which the valence electrons lie in determines the element’s block. As a result, noble gases are also given electronegativity values on this scale.  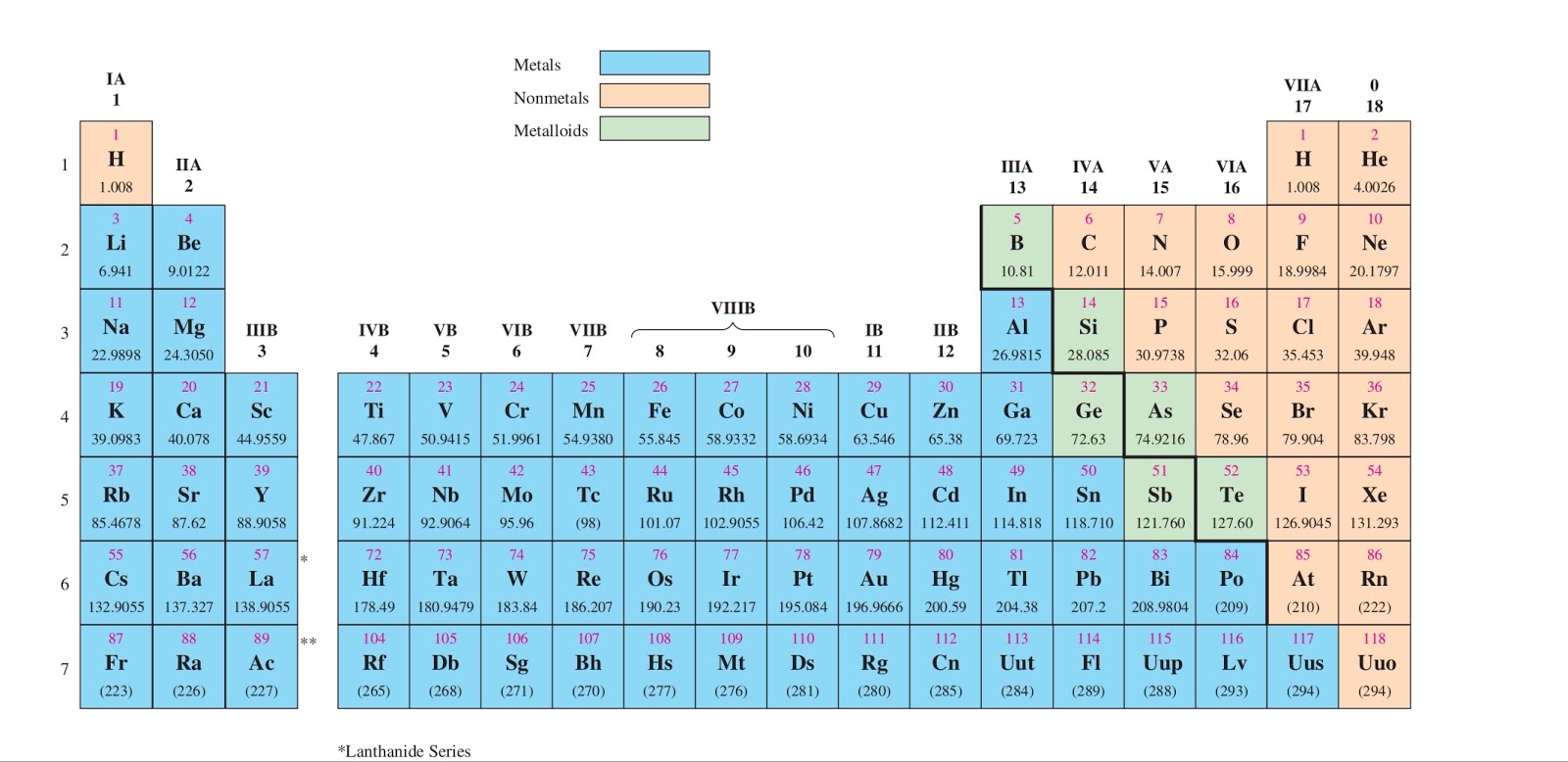
The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. 
If the number is larger than 10, subtract 10 so you get two valence electrons. If you look at the periodic table and at the period numbers, that is the number of valence electrons. \): The Allen electronegativity values of the second-row elements. Valence electrons are the electrons contained in the outermost shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
