|
de Donder, Albert Einstein, John Ellis, Victor Henri, Jacobi, Paul Langevin, Mainfroid, Jean Perrin, Henri Poincare, Erwin Schrodinger, Manne Siegbahn Academie des Sciences, Solvay Congress (1911), Solvay Congress (1927), and SorbonneĪmerican Institute of Physics - Niels Bohr Library and Archivesĭonder, Th.=9. This formulation, then, is called Wave Mechanics. In 1926 Schrdinger published a series of papers giving a full form of Quantum Mechanics in this formulation the central idea is de Broglie's hypothesis. He merged the idea of thinking about them as particles, and of. So that is how de Broglie got his PhD, and in 1926 Davisson and Germer actually saw electrons demonstrating an interference pattern. 
He suggested that an electron around a nucleus could be thought of as being a standing wave, and that electrons and all matter could be considered as waves. Wave-particle duality proposed by de Broglie 1924 complete In 1924, Louis de Broglie proposed that all moving particles, including subatomic particles like electrons, exhibit wave-like behavior. In 1924 Louis de Broglie in his PhD thesis Recherches sur la thorie des quanta introduced his theory of electron waves. 
Also prominently mentioned are: David Bohm, Emile Bourgeois, Leon Brillouin, Marcel Brillouin, Maurice de Broglie, Charles Galton Darwin, Alexandre Dauvillier, Deslandres, Th. A timeline about Atomic Theory, covering key discoveries and advancements in our understanding of the atom. See examples of problems and solutions using his formula. In his 1924 thesis, de Broglie had proposed a theory of. De Broglie equation was the work of Louis de. Immersing himself in an array of theoretical physics research, Schrdinger came upon the work of fellow physicist Louis de Broglie in 1925. For most of his life he believed the former although as a young researcher he had at first believed that the statistics hide our ignorance. Explore his history, equation, and contributions to the Bohr model and the universal atomic theory. The two theories, de Broglie equation and Bohrs hypothesis of atoms, have a great impact on quantum mechanics. The central question in de Broglies life was whether the statistical nature of atomic physics reflects an ignorance of the underlying quantum theory or whether statistics is all that can be known. He took as a starting point the idea that all particles (electrons, light quanta, etc. Learn how French physicist Louis De Broglie discovered the wavelength of electrons and developed his wave-particle theory of matter. Discussions of scientific matters relate to work that was done between approximately 19, with an emphasis on the discovery and interpretations of quantum mechanics in the 1920s. Louis de Broglie’s theory was first presented in a series of papers published in 1923-1924 (Broglie, 1923a, 1923b, 1923c, 1923d 1924a, 1924b, 1924c) and in his PhD thesis (Broglie, 1924d 1925). He introduced his theory of particle-wave. Louis de Broglie has discovered the wave nature of electrons. This concept is known as the de Broglie hypothesis, an example of waveparticle duality, and forms a central part of the theory of quantum mechanics. 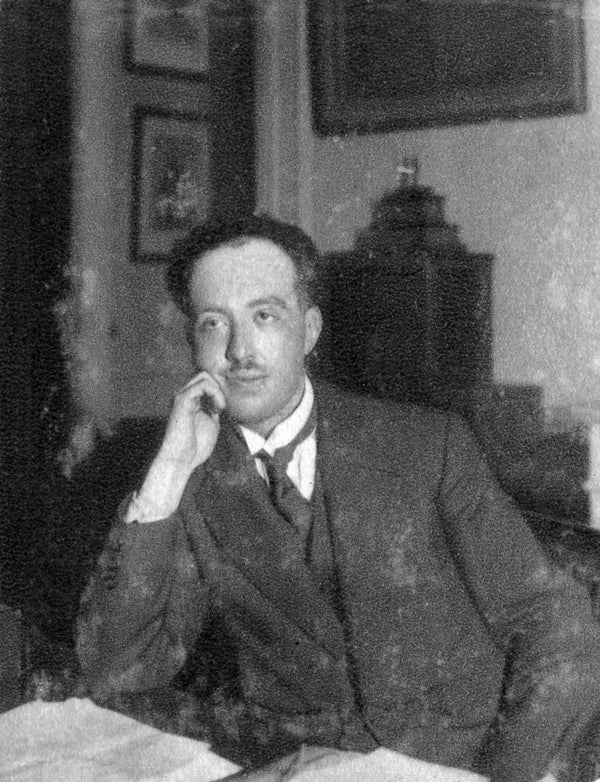
Subjects discuss their family backgrounds, how they became interested in physics, their educations, people who influenced them, their careers including social influences on the conditions of research, and the state of atomic, nuclear, and quantum physics during the period in which they worked. He has uncovered one of the secrets of the universe. Part of the Archives for the History of Quantum Physics oral history collection, which includes tapes and transcripts of oral history interviews conducted with circa 100 atomic and quantum physicists.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
